Life
These Birth Control Pills Were Recalled Over A Packaging Error, So Check Yours ASAP
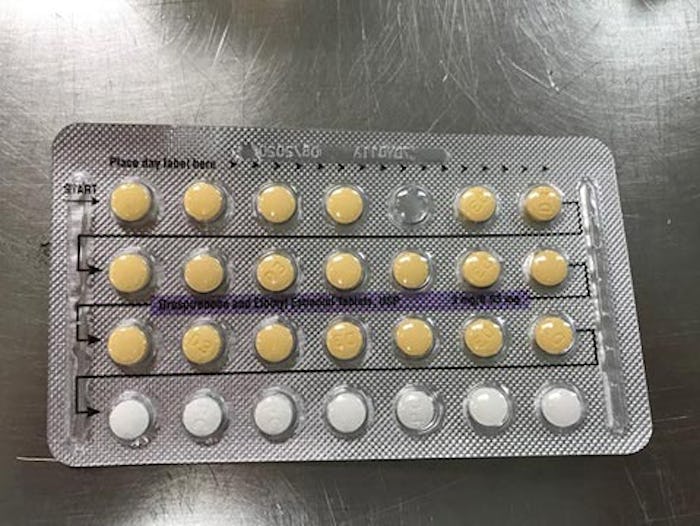
The U.S. Food and Drug Administration (FDA) announced on Monday that a batch of birth control pills has been voluntarily recalled due to packaging error that may lead users to take placebo pills when they're meant to take active pills. Apotex Corp., originally a Canadian pharmaceutical production company with operations in Florida, recalled four lots of Drospirenone and Ethinyl Estradiol Tablets from wholesalers, distributors, and consumers as a result of missing or incorrect tablet arrangements.
“As a result of this packaging error, where a patient does not take a tablet due to a missing tablet or that a patient takes a placebo instead of an active tablet, loss of efficacy is possible due to variation in the dosage consumed,” the FDA said on its website. This means women risk pregnancy if taking the affected products, however, no cases of pregnancy or other adverse effects have yet been reported, the FDA noted.
The following lot numbers have been affected by the defective packaging:
- 7DY008A
- 7DY009A
- 7DY010A
- 7DY011A
Each package contains a strength of 3MG / 0.03MG and notes an expiration date of August 2020, according to the FDA's recall notice. The outer packaging includes the National Drug Code (NDC) number 60505-4183-3 and the NDC on the inside carton is listed as 60505-4183-1.
The products were manufactured by Oman Pharmaceutical Products Co. LLC, under contract from Helm AG, Nordkanalstrasse 28, Hamburg, 20097, Germany. The FDA also provided the images below to help users identify if their birth control is included in the recall.
Apotex has notified affected wholesalers and distributors through mail and patients who have been provided the recalled lots are being urged to contact their pharmacy and health care provider with any questions or concerns. Those with the defective products who purchased directly from Apotex should call 1- 877-674-2082. Consumers with any other questions are being directed to call 1-800-706-5575.
Apotex told Romper in a statement that the incorrect tablet arrangements and empty blister pockets "were identified during an investigational inspection of NON-distributed inventory." The recall, Apotex said, was issued "out of an abundance of caution."
The FDA is also urging those who have experienced adverse reactions or quality problems with the product to report to the FDA’s MedWatch Adverse Event Reporting program online, through regular mail, or through fax to 1-800-FDA-0178.
The agency warned that those using birth control “should not interrupt their therapy” and should “use a non-hormonal method of birth control." One of the most common forms of non-hormonal contraceptives are condoms, according to Planned Parenthood. Other popular alternatives include the diaphragm/cervical cap, intrauterine devices (IUDs), and the sponge.
In 2018, the Centers for Disease Control and Prevention (CDC) reported a shift in contraceptive preference, noting that an increasing number of women prefer longer-lasting reversible birth control methods such as IUDs and contraceptive shots or implants. Here is a good resource for those seeking other forms of contraception.
Last year, Dr. Terri Cornelison, assistant director for the health of women in the FDA's Center for Devices and Radiological Health, warned against the effectiveness of contraception saying "no form of contraception works perfectly." As such, always talk to your health care provider for the best form of contraception fit for your needs.
Although there have been zero reported pregnancies or other adverse affects resulted from the packaging error, be sure to double-check your birth control tablets for missing or incorrect pills. And if you have any other concerns, call your physician or pharmacy ASAP for alternative options.